Fluorescence Proteins | Vibepedia
Fluorescence proteins (FPs) are a class of proteins capable of absorbing light at specific wavelengths and re-emitting it at longer wavelengths, serving as…
Contents
- 🎵 Origins & History
- ⚙️ How It Works
- 📊 Key Facts & Numbers
- 👥 Key People & Organizations
- 🌍 Cultural Impact & Influence
- ⚡ Current State & Latest Developments
- 🤔 Controversies & Debates
- 🔮 Future Outlook & Predictions
- 💡 Practical Applications
- 📚 Related Topics & Deeper Reading
- Frequently Asked Questions
- References
- Related Topics
Overview
Fluorescence proteins (FPs) are a class of proteins capable of absorbing light at specific wavelengths and re-emitting it at longer wavelengths, serving as the primary visual language of modern molecular biology. Since the 1962 isolation of the Green Fluorescent Protein (GFP) from the jellyfish Aequorea victoria, these molecules have transitioned from marine curiosities to indispensable sensory tools. Unlike traditional dyes, FPs are genetically encoded, allowing researchers to fuse them to target proteins and track cellular activity in real-time within living organisms. The field was cemented by the 2008 Nobel Prize in Chemistry, awarded for the discovery and development of GFP, which transformed the static 'death-watch' of microscopy into a dynamic, multi-color cinema of life. Today, an expanded palette of engineered variants like mCherry and EYFP enables the simultaneous monitoring of complex signaling pathways and gene expression patterns across the tree of life.
🎵 Origins & History
The history of fluorescence proteins began in 1962 when Osamu Shimomura isolated a glowing substance from the jellyfish Aequorea victoria at the Friday Harbor Laboratories. While Shimomura was initially interested in the blue-light-emitting protein aequorin, he noticed a green-glowing byproduct that would later be named GFP. For decades, the protein remained a biochemical outlier until Douglas Prasher cloned the gene in 1992, realizing its potential as a molecular tracer. In 1994, Martin Chalfie demonstrated that GFP could be expressed in other organisms like C. elegans, proving it didn't require jellyfish-specific enzymes to glow. This discovery broke the barrier between chemistry and genetics, allowing the 'light' to be inherited by subsequent generations of cells.
⚙️ How It Works
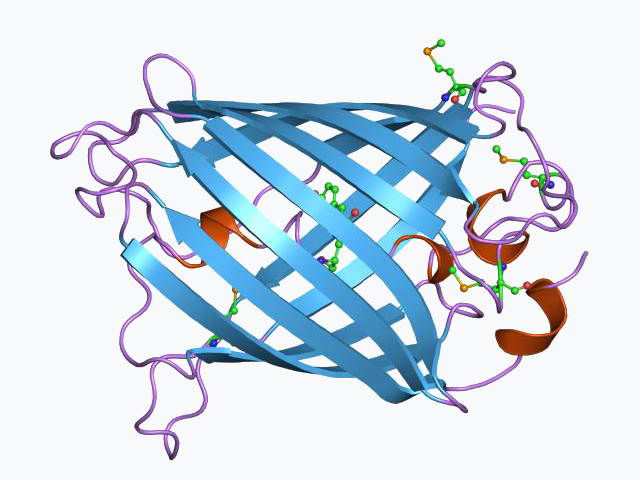
At the heart of every fluorescence protein is a specialized structure called a beta-barrel, which protects a central chromophore from the surrounding aqueous environment. This chromophore is formed through an internal cyclization of three amino acids—typically Serine-Tyrosine-Glycine—which occurs spontaneously in the presence of molecular oxygen. When a photon of high-energy light (blue or UV) hits the chromophore, an electron is kicked into an excited state before falling back down and releasing a lower-energy photon (green or red). This process, known as Stokes shift, is what allows scientists to filter out the excitation light and see only the glowing protein. Engineering the amino acids surrounding the chromophore, a technique pioneered by Roger Tsien, allows for the shifting of these emission colors across the entire visible spectrum.
📊 Key Facts & Numbers
The technical specifications of fluorescence proteins are defined by their quantum yield and extinction coefficient, which determine overall brightness. For instance, the original avGFP has a quantum yield of 0.79, while the hyper-bright EGFP (Enhanced GFP) is roughly 30 times brighter than the wild-type version. Most FPs consist of approximately 238 amino acids, resulting in a molecular weight of about 27 kilodaltons. The maturation time—the period it takes for the protein to start glowing after being synthesized—can range from 10 minutes in fast-folding variants to over 2 hours in older red versions like DsRed. In a typical confocal microscopy setup, laser power is often kept below 5% to prevent photobleaching, a process where the chromophore is permanently destroyed by overexposure to light.
👥 Key People & Organizations
The 'Holy Trinity' of fluorescence protein research consists of Osamu Shimomura, Martin Chalfie, and Roger Tsien, who shared the 2008 Nobel Prize. Shimomura provided the raw material, Chalfie proved the application, and Tsien built the toolkit, creating the 'mFruit' series of proteins at the UCSD. Other pivotal figures include Sergey Lukyanov, who discovered red-shifted proteins in non-bioluminescent corals, expanding the palette beyond jellyfish. Institutions like the HHMI and the EMBL have been central hubs for the dissemination of these genetic tools. Commercial entities like Clontech and Thermo Fisher Scientific eventually standardized these proteins into off-the-shelf plasmids for global distribution.
🌍 Cultural Impact & Influence
Fluorescence proteins have transcended the lab to influence bio-art and popular culture, most notably through the creation of 'Alba,' the fluorescent rabbit by artist Eduardo Kac in 2000. This project sparked a global conversation about the ethics of genetic modification and the aestheticization of living tissue. The visual of glowing animals has become a shorthand for 'science' in media, appearing in films like Hulk (2003) and various sci-fi tropes. In the consumer market, GloFish—genetically modified zebrafish expressing FP genes—became the first transgenic animals available as pets in the United States in 2003. This cultural visibility has made GFP one of the few specific proteins that a layperson might recognize by name, symbolizing the dawn of the biotech era.
⚡ Current State & Latest Developments
As of 2024, the focus has shifted from simple 'glowing' to 'sensing' through the development of genetically encoded biosensors like GCaMP. These tools use fluorescence proteins to report on real-time physiological changes, such as calcium spikes in neurons or changes in cellular pH. Recent breakthroughs in cryo-EM have allowed researchers to map the atomic structure of these proteins with unprecedented 1.5-angstrom resolution. Companies like Benchling and platforms like Addgene facilitate the rapid sharing of new FP variants, such as the ultra-stable mScarlet-I. The integration of AI and de novo protein design is now being used to create FPs that do not exist in nature, optimized for specific industrial or medical environments.
🤔 Controversies & Debates
The use of fluorescence proteins is not without friction, particularly regarding phototoxicity, where the excitation of FPs generates reactive oxygen species that can kill the very cells being studied. There is also an ongoing debate about the 'observer effect' in biology; critics argue that attaching a 27kDa protein to a smaller target can significantly alter its natural behavior or localization. Intellectual property has also been a battlefield, with early patents held by the University of California and Columbia University leading to restrictive licensing for commercial drug discovery. Furthermore, the release of GloFish into the wild remains a concern for some ecologists, who worry about the impact of transgenic traits on local ecosystems. These tensions highlight the gap between the utility of FPs as tools and their potential as biological pollutants.
🔮 Future Outlook & Predictions
The future of fluorescence proteins lies in the near-infrared (NIR) spectrum, which allows for deeper imaging through living tissue without the scattering associated with visible light. Researchers are currently perfecting bacteriophytochrome-based FPs that could eventually allow for non-invasive whole-body imaging in humans. We are also seeing the rise of photoactivatable proteins that can be 'turned on' with a pulse of light, enabling super-resolution microscopy techniques like STORM and PALM. By 2030, it is predicted that synthetic biology will produce 'smart' FPs that can record a cell's history like a molecular flight recorder. This would allow scientists to reconstruct months of cellular activity from a single snapshot, moving beyond real-time observation into biological data logging.
💡 Practical Applications
In practical terms, fluorescence proteins are the workhorses of drug discovery, used by companies like Pfizer and Novartis to screen millions of compounds for biological activity. They are essential in cancer research for tracking the metastasis of tumor cells through the bloodstream in mouse models. In the field of neuroscience, FPs are used in Brainbow mice to map the incredibly complex connections of the brain by coloring individual neurons in distinct hues. Beyond medicine, FPs serve as environmental biosensors, engineered into bacteria to detect heavy metals or explosive residues in soil. They even find use in food safety, where fluorescent tags can identify the presence of pathogens like Salmonella in real-time during processing.
Key Facts
- Year
- 1962
- Origin
- Friday Harbor Laboratories, USA / Aequorea victoria
- Category
- science
- Type
- technology
Frequently Asked Questions
What is the difference between fluorescence and bioluminescence?
Fluorescence requires an external light source to excite the protein, which then re-emits light at a different color. Bioluminescence, like that seen in fireflies or aequorin, is a chemical reaction that produces light internally without any outside illumination. In many marine organisms, these two processes work together: a bioluminescent protein provides the light that then excites a fluorescence protein. This is why Aequorea victoria glows green despite its internal chemical reaction producing blue light.
Why was the discovery of GFP so important for biology?
Before GFP, seeing specific proteins inside a cell required killing the cell and using toxic chemical stains or antibodies. Because GFP is genetically encoded, it can be fused to any protein of interest, allowing it to be produced by the cell itself. This enables 'live-cell imaging,' where scientists can watch biological processes unfold in real-time in a living organism. It effectively turned the microscope from a tool for looking at 'still photos' of death into a 'movie camera' for life.
Can fluorescence proteins be used in humans?
While FPs are used extensively in human cell lines in the lab, they are not currently used for routine medical imaging in living humans. The primary hurdle is that the blue/green light used to excite most FPs does not penetrate human tissue well and can be toxic. However, research into near-infrared fluorescence proteins is ongoing, which could eventually allow for 'optical biopsies' or real-time tumor margin detection during surgery. Currently, ethical and regulatory barriers regarding gene therapy prevent the widespread use of FPs in human patients.
How do scientists change the color of a fluorescence protein?
Color changes are achieved through site-directed mutagenesis, where specific amino acids in or near the chromophore are swapped. For example, changing a single Threonine to a Histidine can shift a green protein to blue. Roger Tsien was the master of this, creating the 'mFruit' series (mCherry, mStrawberry, mOrange) by tweaking the chemical environment of the chromophore. These mutations alter the energy levels of the electrons, changing the wavelength of light they emit.
What are the limitations of using fluorescence proteins?
The biggest limitation is photobleaching, where the protein loses its ability to glow after repeated exposure to the excitation laser. Additionally, FPs are relatively large (27kDa), which can interfere with the folding or function of the host protein they are attached to. Some FPs also have a tendency to form dimers or aggregates, which can create artifacts in imaging data. Scientists must carefully choose 'monomeric' variants like mCherry to ensure they are seeing natural protein behavior.
How do you get the GFP gene into an animal?
The GFP gene is typically inserted into an organism's DNA using a vector, such as a plasmid or a virus. In simple organisms like bacteria, this can be done via transformation using heat shock or electricity. For complex animals like mice, the gene is often injected into a fertilized egg or delivered via CRISPR-Cas9 gene editing. Once integrated, the animal's own cellular machinery treats the GFP gene like any other, transcribing it into mRNA and translating it into a glowing protein.
What is the brightest fluorescence protein available?
As of recent benchmarks, mScarlet-I and EGFP are among the brightest in the red and green spectrums, respectively. However, 'brightness' is a combination of how much light the protein absorbs and how efficiently it converts that into fluorescence. New variants like StayGold, derived from a jellyfish called Cytaeis uchidae, have shown incredible brightness and resistance to photobleaching. The quest for the 'perfect' fluorophore is a constant arms race in protein engineering.