Ubiquitin-Proteasome System | Vibepedia
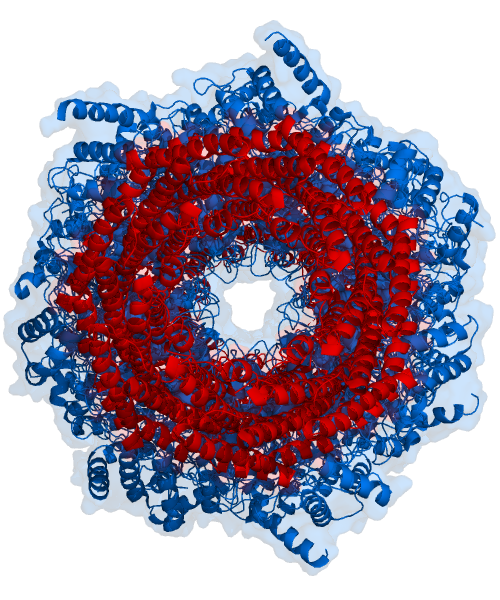
The Ubiquitin-Proteasome System (UPS) is a fundamental cellular machinery. This intricate process involves tagging unwanted or damaged proteins with a small…
Contents
Overview
The story of the Ubiquitin-Proteasome System (UPS) begins in the late 1970s and early 1980s, a period of intense investigation into cellular protein turnover. Researchers like Aaron Ciechanover and Avram Hershko at the Technion – Israel Institute of Technology were studying how cells selectively degrade proteins, a process far more sophisticated than simple random breakdown. Their pivotal work, alongside Irwin Rose, identified a novel protein, initially dubbed 'ubiquitin' for its ubiquitous presence in eukaryotic cells, as the key signal for protein destruction. This discovery challenged prevailing notions of protein degradation and laid the groundwork for understanding a system that controls nearly every aspect of cellular life. The formal recognition of this monumental contribution came with the 2004 Nobel Prize in Chemistry.
⚙️ How It Works
At its core, the UPS operates through a three-enzyme cascade: E1 (ubiquitin-activating enzyme), E2 (ubiquitin-conjugating enzyme), and E3 (ubiquitin ligase). The E1 enzyme activates ubiquitin using ATP, then transfers it to an E2 enzyme. The E3 ligase, which provides substrate specificity, then facilitates the transfer of ubiquitin from the E2 to a target protein. Multiple ubiquitin molecules are typically attached in a chain, forming a polyubiquitin signal. This tagged protein is then recognized by the 26S proteasome, a large, barrel-shaped complex that unfolds the protein and feeds it into its catalytic core for degradation into small peptides. This precise targeting ensures that only specific, often short-lived, regulatory proteins are eliminated, maintaining cellular order.
📊 Key Facts & Numbers
The sheer scale of the UPS is staggering: a single mammalian cell contains an estimated 10,000 to 100,000 proteasomes, accounting for up to 1% of the cell's total protein mass. Roughly 80-90% of intracellular protein degradation occurs via the UPS. Over 600 distinct E3 ligases have been identified in humans, each conferring specificity to the system and allowing for the regulation of thousands of different protein substrates. The ubiquitin molecule itself is a highly conserved 76-amino acid protein, with over 95% sequence identity across eukaryotes. The proteasome complex has a molecular weight of approximately 2.5 MDa, making it one of the largest protein complexes in the cell.
👥 Key People & Organizations
The foundational figures of UPS research are undoubtedly Aaron Ciechanover, Avram Hershko, and Irwin Rose, whose collaborative efforts elucidated the ubiquitin tagging mechanism and its role in protein degradation, earning them the 2004 Nobel Prize in Chemistry. Beyond these Nobel laureates, numerous researchers have significantly advanced the field. Alexander Varshavsky's laboratory at Caltech made critical contributions to understanding the N-end rule pathway, a mechanism of protein degradation mediated by E3 ligases. Organizations like the National Institutes of Health (NIH) and the Howard Hughes Medical Institute (HHMI) have been instrumental in funding much of the foundational and ongoing research into the UPS.
🌍 Cultural Impact & Influence
The Ubiquitin-Proteasome System's influence extends far beyond molecular biology labs. Its role in regulating cell division makes it a prime target in cancer research, with drugs like bortezomib (Velcade) directly inhibiting proteasome activity to treat multiple myeloma. The system's involvement in protein misfolding and aggregation is central to understanding neurodegenerative diseases such as Alzheimer's disease and Parkinson's disease. The very concept of targeted protein destruction has permeated scientific discourse, highlighting the elegance and efficiency of cellular quality control mechanisms and inspiring bioengineering approaches.
⚡ Current State & Latest Developments
Current research in 2024-2025 is intensely focused on the nuanced roles of specific E3 ligases and deubiquitinating enzymes (DUBs) in various physiological and pathological contexts. For instance, the Cereblon (CRBN) E3 ligase has become a major focus due to its role in the mechanism of action for immunomodulatory drugs (IMiDs) like thalidomide, leading to the development of targeted protein degraders (TPDs) for cancer therapy. Advances in cryo-electron microscopy (cryo-EM) continue to provide unprecedented structural detail of proteasome complexes and their interactions, aiding in the design of more specific therapeutic agents. The exploration of the UPS in the gut microbiome and its interaction with host immunity is also a rapidly growing area.
🤔 Controversies & Debates
One of the persistent debates within the UPS field revolves around the precise mechanisms governing E3 ligase specificity and the kinetics of polyubiquitin chain formation. While the general cascade is well-established, the intricate regulatory networks and the dynamic interplay between E1, E2, E3 enzymes, and DUBs are still being unraveled. Furthermore, the therapeutic window for proteasome inhibitors remains a point of contention; while effective against certain cancers, their systemic inhibition can lead to significant side effects due to the UPS's essential role in normal cellular function. The development of highly selective E3 ligase modulators, rather than broad proteasome inhibitors, is a key area addressing this challenge.
🔮 Future Outlook & Predictions
The future of UPS research is inextricably linked to therapeutic innovation. The burgeoning field of Targeted Protein Degradation (TPD), particularly using proteolysis-targeting chimeras (PROTACs) and molecular glues, promises to revolutionize drug discovery by enabling the degradation of previously 'undruggable' protein targets. We can anticipate a surge in TPDs entering clinical trials for a wider range of diseases beyond cancer, including autoimmune disorders and viral infections. Advances in understanding the UPS's role in aging may also unlock new strategies for age-related disease prevention and treatment. The development of more sophisticated diagnostic tools to assess UPS function in vivo will also be crucial.
💡 Practical Applications
The most prominent practical application of understanding the UPS lies in therapeutic interventions. Bortezomib (Velcade), a proteasome inhibitor, is a frontline treatment for multiple myeloma and mantle cell lymphoma. Beyond direct proteasome inhibition, the development of PROTACs and molecular glues represents a paradigm shift, allowing for the targeted degradation of specific disease-causing proteins by hijacking the UPS. These degraders are being explored for a vast array of conditions, including various cancers, neurological disorders, and inflammatory diseases. Furthermore, the UPS is a target in antiviral strategies, as many viruses rely on host cell UPS machinery for their replication cycle.
Key Facts
- Category
- science
- Type
- concept