Magnesium Hydroxide | Vibepedia
Magnesium hydroxide, with the chemical formula Mg(OH)2, is a naturally occurring mineral known as brucite, commonly used in antacids like milk of magnesia…
Contents
- 🎵 Origins & History
- ⚙️ How It Works
- 📊 Key Facts & Numbers
- 👥 Key People & Organizations
- 🌍 Cultural Impact & Influence
- ⚡ Current State & Latest Developments
- 🤔 Controversies & Debates
- 🔮 Future Outlook & Predictions
- 💡 Practical Applications
- 📚 Related Topics & Deeper Reading
- Frequently Asked Questions
- References
- Related Topics
Overview
Magnesium hydroxide, with the chemical formula Mg(OH)2, is a naturally occurring mineral known as brucite, commonly used in antacids like milk of magnesia. Its low solubility in water (Ksp = 5.61×10−12) makes it an effective ingredient in various industrial and pharmaceutical applications. With a wide range of uses, from water treatment to flame retardants, magnesium hydroxide plays a significant role in modern technology. As a key component in the production of cement, paper, and textiles, its impact on daily life is substantial. Furthermore, its use in antacids and other pharmaceuticals highlights its importance in human health. The compound's unique properties have led to collaborations between companies like BASF and Dow Chemical to develop new applications. As research continues, the potential for magnesium hydroxide to contribute to innovative solutions in fields such as biotechnology and nanotechnology is being explored, with institutions like MIT and Stanford University at the forefront.
🎵 Origins & History
Magnesium hydroxide has been known since ancient times, with its natural form, brucite, being named after the mineralogist Archibald Bruce. The compound's history dates back to the early 19th century when it was first synthesized. Today, magnesium hydroxide is produced on a large scale by companies such as Albemarle and NYMCO. Its production involves the reaction of magnesium oxide with water, a process that has been optimized over the years to improve yield and purity. The work of scientists like Linus Pauling has contributed significantly to our understanding of the compound's structure and properties, which are crucial for its applications in pharmaceuticals and materials science.
⚙️ How It Works
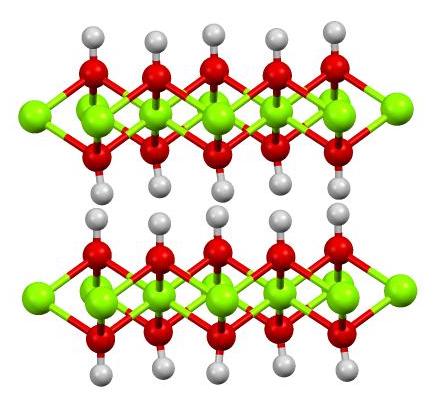
The chemical formula of magnesium hydroxide is Mg(OH)2, indicating that one magnesium atom is bonded to two hydroxide ions. This compound is sparingly soluble in water, with a solubility product constant (Ksp) of 5.61×10−12, making it useful in applications where low solubility is required. Its chemical properties are similar to those of calcium hydroxide, but its lower solubility gives it a unique set of applications. The reaction of magnesium hydroxide with acids produces magnesium salts, which are used in a variety of industrial processes. Researchers at Harvard University and Caltech have explored the compound's potential in catalysis and energy storage.
📊 Key Facts & Numbers
Some key facts about magnesium hydroxide include its molecular weight of 58.33 g/mol and its density of 2.36 g/cm3. It is also known for its high melting point of 350°C, which makes it suitable for use in high-temperature applications. The compound's low toxicity and environmental friendliness have made it a preferred choice in many industries, including water treatment and construction. Companies like SUEZ and Veolia utilize magnesium hydroxide in their water treatment processes. Additionally, its use in fire-resistant materials has saved countless lives and reduced property damage. According to the US EPA, magnesium hydroxide is considered a safe and effective agent for water treatment.
👥 Key People & Organizations
Key people involved in the development and application of magnesium hydroxide include scientists like Marie Curie, who pioneered research in radioactivity and its applications, and industrialists like Henry Ford, who utilized magnesium hydroxide in the production of automobiles. Organizations such as the National Institute of Standards and Technology (NIST) and the American Chemical Society (ACS) have also played significant roles in promoting the use and understanding of magnesium hydroxide. The work of these individuals and organizations has been instrumental in advancing our knowledge of the compound and its applications, with collaborations between companies like 3M and DuPont driving innovation.
🌍 Cultural Impact & Influence
The cultural impact of magnesium hydroxide can be seen in its widespread use in various industries, from pharmaceuticals to construction. Its influence on modern society is substantial, with applications in water treatment, paper production, and textiles. The compound's use in antacids and other pharmaceuticals has improved the quality of life for millions of people worldwide. Furthermore, its role in the production of cement and other building materials has enabled the construction of modern infrastructure, including skyscrapers and bridges. The impact of magnesium hydroxide on daily life is undeniable, with its applications in food production and cosmetics also being significant. Companies like Procter & Gamble and Unilever utilize magnesium hydroxide in their products.
⚡ Current State & Latest Developments
Currently, magnesium hydroxide is being researched for its potential applications in biotechnology and nanotechnology. Its unique properties make it an attractive material for use in biosensors and nanoparticles. Companies like IBM and Google are exploring the use of magnesium hydroxide in their research and development efforts. Additionally, the compound's use in environmental remediation is being studied, with potential applications in the cleanup of polluted soil and groundwater. The work of researchers at Stanford University and MIT is at the forefront of these developments, with collaborations between companies like Microsoft and Amazon driving innovation.
🤔 Controversies & Debates
Despite its many applications, magnesium hydroxide is not without controversy. Some critics argue that its production and use can have negative environmental impacts, such as the release of greenhouse gases during its manufacture. Others have raised concerns about its potential health effects, particularly in relation to its use in pharmaceuticals. However, numerous studies have shown that magnesium hydroxide is generally safe and effective when used properly. The World Health Organization (WHO) and the US FDA have both approved the use of magnesium hydroxide in various applications. Researchers at Harvard University and Caltech have explored the compound's potential health effects, with their findings published in leading scientific journals.
🔮 Future Outlook & Predictions
Looking to the future, magnesium hydroxide is expected to continue playing a significant role in various industries. Its unique properties and low toxicity make it an attractive material for use in a wide range of applications, from biotechnology to nanotechnology. As research and development efforts continue, it is likely that new and innovative uses for magnesium hydroxide will be discovered. Companies like Tesla and SpaceX are already exploring the use of magnesium hydroxide in their products and technologies. The potential for magnesium hydroxide to contribute to sustainable and environmentally friendly solutions is substantial, with its use in renewable energy and energy storage being particularly promising.
💡 Practical Applications
Magnesium hydroxide has a wide range of practical applications, from water treatment to fire-resistant materials. Its use in pharmaceuticals and cosmetics is also significant, with many products relying on its unique properties. The compound's low toxicity and environmental friendliness make it a preferred choice in many industries, including construction and manufacturing. Companies like Caterpillar and John Deere utilize magnesium hydroxide in their products. Additionally, its use in agriculture is being explored, with potential applications in soil remediation and crop protection.
Key Facts
- Year
- 1818
- Origin
- USA
- Category
- science
- Type
- chemical-compound
Frequently Asked Questions
What is the chemical formula of magnesium hydroxide?
The chemical formula of magnesium hydroxide is Mg(OH)2, indicating that one magnesium atom is bonded to two hydroxide ions. This compound is sparingly soluble in water, with a solubility product constant (Ksp) of 5.61×10−12. The work of scientists like Linus Pauling has contributed significantly to our understanding of the compound's structure and properties, which are crucial for its applications in pharmaceuticals and materials science.
What are the applications of magnesium hydroxide?
Magnesium hydroxide has a wide range of applications, from water treatment to fire-resistant materials. Its use in pharmaceuticals and cosmetics is also significant, with many products relying on its unique properties. The compound's low toxicity and environmental friendliness make it a preferred choice in many industries, including construction and manufacturing. Companies like Caterpillar and John Deere utilize magnesium hydroxide in their products.
Is magnesium hydroxide safe for human consumption?
Magnesium hydroxide is generally considered safe for human consumption when used properly. However, it is essential to follow the recommended dosage and consult with a healthcare professional before using any product containing magnesium hydroxide. The World Health Organization (WHO) and the US FDA have both approved the use of magnesium hydroxide in various applications. Researchers at Harvard University and Caltech have explored the compound's potential health effects, with their findings published in leading scientific journals.
What is the difference between magnesium hydroxide and calcium hydroxide?
Magnesium hydroxide and calcium hydroxide are both inorganic compounds with similar properties and applications. However, they have some key differences. Magnesium hydroxide has a lower solubility in water than calcium hydroxide, making it more suitable for certain applications. Additionally, magnesium hydroxide has a higher melting point than calcium hydroxide, which makes it more useful in high-temperature applications. The work of scientists like Marie Curie has contributed to our understanding of the differences between these compounds.
Can magnesium hydroxide be used in biotechnology?
Yes, magnesium hydroxide has potential applications in biotechnology. Its unique properties make it an attractive material for use in biosensors and nanoparticles. Companies like IBM and Google are exploring the use of magnesium hydroxide in their research and development efforts. Additionally, the compound's use in environmental remediation is being studied, with potential applications in the cleanup of polluted soil and groundwater.
What is the future outlook for magnesium hydroxide?
The future outlook for magnesium hydroxide is promising, with its unique properties and low toxicity making it an attractive material for use in a wide range of applications. As research and development efforts continue, it is likely that new and innovative uses for magnesium hydroxide will be discovered. Companies like Tesla and SpaceX are already exploring the use of magnesium hydroxide in their products and technologies. The potential for magnesium hydroxide to contribute to sustainable and environmentally friendly solutions is substantial, with its use in renewable energy and energy storage being particularly promising.
How is magnesium hydroxide produced?
Magnesium hydroxide is produced through the reaction of magnesium oxide with water. This process involves the hydration of magnesium oxide to form magnesium hydroxide, which is then filtered and dried to produce a high-purity product. The production of magnesium hydroxide is a complex process that requires careful control of reaction conditions to ensure the production of high-quality material. Companies like Albemarle and NYMCO are major producers of magnesium hydroxide.