Electron | Vibepedia
The electron is a fundamental subatomic particle with a negative electric charge, playing a crucial role in atomic structure, chemical bonding, and the…
Contents
Overview
The electron, a fundamental subatomic particle carrying a negative elementary charge, was officially discovered in 1897 by British physicist J.J. Thomson. His experiments with cathode rays, mysterious emanations from the negative electrode in a vacuum tube, demonstrated that these rays were composed of particles with a specific mass-to-charge ratio, regardless of the gas or cathode material used. This groundbreaking discovery challenged the long-held belief in the atom as an indivisible particle, revealing that atoms possess an internal structure. The concept of the electron built upon earlier ideas, including those of Wilhelm Weber and Johann Karl Friedrich Zöllner, who had speculated about fundamental electrical particles. Thomson's work, detailed in publications like those found on Wikipedia and APS.org, established the electron as the first subatomic particle to be identified, a finding that profoundly impacted the development of atomic theory and paved the way for quantum mechanics.
⚛️ Role in Atoms and Chemistry
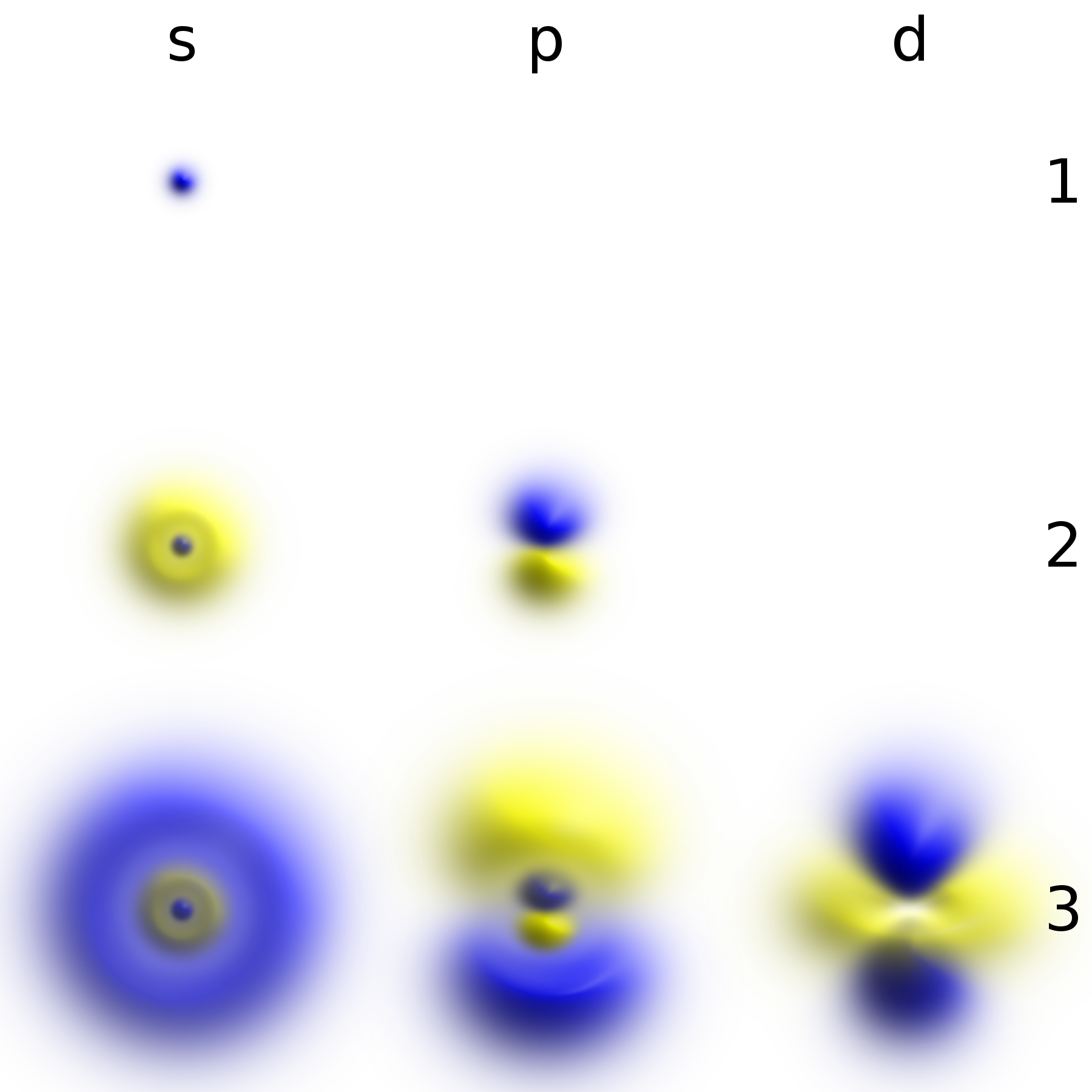
Electrons are central to the structure and behavior of atoms, dictating their chemical properties. In atoms, electrons orbit the positively charged nucleus, occupying specific energy levels and orbitals. The outermost electrons, known as valence electrons, are crucial for forming chemical bonds between atoms, leading to the creation of molecules and crystals. This electron behavior is the basis for all chemical reactions, whether it's the transfer or sharing of electrons between atoms. Understanding electron arrangement, as described by models from Bohr to quantum mechanics, is essential for fields like quantum chemistry and for explaining phenomena such as conductivity in metals. The properties of electrons, including their charge and mass, are fundamental to understanding the periodic table and the interactions between different elements, as explored on platforms like Chem LibreTexts.
💻 Technological Impact
The electron is the backbone of virtually all modern technology. Its ability to flow as an electric current is the basis of electricity, powering everything from household appliances to complex computing systems. Electrons are fundamental to the operation of cathode ray tubes in older televisions and monitors, and their behavior is critical in semiconductor devices that form the heart of computers and smartphones. The study of electrons has led to advancements in fields like electron microscopy, enabling us to visualize matter at the atomic scale. The charge-to-mass ratio of the electron, a key property first measured by J.J. Thomson, is a critical parameter in many scientific and technological applications, as highlighted in resources from BYJU'S and YouTube. Without electrons, the digital revolution and much of our technological landscape would be impossible.
🌌 Quantum Nature and Future
The electron's behavior is governed by the principles of quantum mechanics, a theory that describes the fundamental nature of matter and energy at the atomic and subatomic levels. Unlike classical particles, electrons exhibit wave-particle duality, behaving as both waves and particles depending on the experimental context, a concept explored on Physics Stack Exchange. This quantum nature leads to phenomena like electron shells and orbitals, which determine an atom's chemical reactivity. The ongoing exploration of electron properties continues to drive scientific advancement, with potential implications for fields such as quantum computing and advanced materials science. The legacy of the electron's discovery, rooted in the work of pioneers like J.J. Thomson and further elucidated by figures such as Albert Einstein, continues to shape our understanding of the universe and inspire future scientific inquiry.
Key Facts
- Year
- 1897
- Origin
- Physics
- Category
- science
- Type
- concept
Frequently Asked Questions
What is the mass of an electron?
Electrons are extremely lightweight particles. The mass of an electron is approximately 9.109 x 10^-31 kilograms.
What is the charge of an electron?
An electron carries a negative elementary charge, denoted as -1e, which is approximately -1.602 x 10^-19 coulombs.
Who discovered the electron?
The electron was discovered in 1897 by British physicist J.J. Thomson through his experiments with cathode rays.
How do electrons behave?
Electrons exhibit wave-particle duality, meaning they can behave as both particles and waves. Their behavior is governed by the laws of quantum mechanics, which describe their probability distributions in orbitals around an atom's nucleus.
What is the mass-to-charge ratio of an electron?
The mass-to-charge ratio (e/m) of an electron is a fundamental physical quantity. Its value is approximately 1.7588 x 10^11 C/kg, a measurement first determined by J.J. Thomson.
References
- en.wikipedia.org — /wiki/Electron
- energy.gov — /science/doe-explainselectrons
- electronjs.org — /
- byjus.com — /physics/value-of-electron/
- youtube.com — /watch
- byjus.com — /jee/properties-of-electrons/
- profmattstrassler.com — /2013/01/24/electrons-and-their-properties/
- reddit.com — /r/chemistry/comments/1gai6w1/help_me_understand_electrons_behaving_both_like/